 | New Jersey Society of Interventional Pain PhysiciansAn Affiliate of the American Society of Interventional Pain Physicians |
in the news
NJSIPP mild Position Statement
-
Date: May 20th, 2026
Re: Coverage Consideration for the mild® Procedure for Lumbar Spinal Stenosis with Neurogenic Claudication
On behalf of the leadership and membership of the New Jersey Society of Interventional Pain Physicians (NJSIPP), we respectfully submit this position statement in support of the mild® Procedure for patients suffering from lumbar spinal stenosis (LSS) with associated neurogenic claudication (NC) and hypertrophic ligamentum flavum (HLF). This endorsement is grounded in robust clinical evidence, FDA clearance, real-world outcomes, and broad utilization across interventional pain practices. We urge payors to adopt favorable coverage policies aligned with the newly approved Category I CPT® codes, effective January 1, 2026, to ensure equitable access to this safe, effective, and minimally invasive therapy.
Lumbar Spinal Stenosis Overview
LSS is a condition in which there is a narrowing in the lumbar spine for neural and vascular structures. This is typically due to age-related degenerative changes in the spinal canal such as HLF, disc herniation, facet hypertrophy, and lateral recess/foraminal narrowing. This often manifests in symptoms which can profoundly affect a patient’s quality of life (QoL), such as pain, tingling, numbness, and weakness in the legs and buttocks. Function can be greatly reduced in the LSS patient, able to walk only a couple hundred feet or stand for less than 10 minutes without experiencing pain.1 Standing and walking will often exacerbate the NC symptoms and forward flexion, sitting, or reliance on walking aids will typically provide relief. The prevalence of LSS is reported to be as high as 8% of the general US population;2 however, in those 60 years of age or older, the prevalence is approximately 20%.3 A cross-sectional population-based cohort study utilizing data from the Framingham Heart Study found the prevalence of LSS to be 8.4%. Acquired LSS was found in 7.3% of patients, while congenital LSS is less common at a prevalence of 2.6%.3 mild® Procedure Overview The mild procedure is a minimally invasive method to restore space in the spinal canal, This procedure is typically conducted on an outpatient basis, through an incision just 5.1mm wide. The mild procedure technology includes a device kit containing specialized surgical instruments intended to perform lumbar decompressive procedures for the treatment of LSS, with HLF as a contributing factor and NC as the presenting symptom. The mild procedure is designed to access the interlaminar space from the posterior lumbar spine, enabling the user to remove small portions of the lamina and resect and debulk the thickened ligamentum flavum accomplishing direct lumbar decompression, under fluoroscopic
guidance. The procedure does not require general anesthesia, stitches, implants, steroids, or opioids. Patients generally return to daily activities within 24 hours without any limitations.4
mild® Procedure Clinical Evidence
By restoring space in the spinal canal, the mild procedure enables patients to experience less discomfort while standing longer and walking greater distances.1 When combined with conventional medical management such as physical therapy, home exercise, walking aids, early interventional therapies such as epidural steroid injections, lumbar facet medical branch nerve blocks, facet joint injections, and radiofrequency ablation, the mild procedure has demonstrated superior outcomes in providing improved function and decreased pain when compared to conventional medical management alone.5 With a safety profile similar to epidural steroid injections (ESIs),6 the mild procedure's effects are proven to last for up to five years.7 Over 110,000 patients across the country have benefited from this procedure, which is backed by more than 30 peer-reviewed articles and 17 clinical studies, including two multi-center randomized controlled trials providing Level 1 evidence. The LSS treatment algorithm positions mild as the next step after conservative care such as rest, physical therapy, ESIs, opioids, etc. and before more invasive implants or surgery.8
Cost Effectiveness
The mild procedure is less costly than surgical spinal procedures and is the best choice in terms of cost and benefits per quality-adjusted life-year (QALY) compared to ESIs and decompression laminectomy.9 Patients also experience a reduction in health care resource utilization after mild, with reductions in specialty care and interventional pain procedures.10
Patient Selection
Identifying the appropriate patient population is essential to optimizing outcomes with mild. Candidates are evaluated based on both symptom presentation and imaging criteria to ensure treatment is targeted to those most likely to benefit. The following clinical and radiographic factors outline the typical patient profile considered for this intervention:
• Pain, numbness, tingling, or heaviness in back, legs, or buttocks when standing or walking.8
• LSS at levels L1-S1 secondary to HLF ≥ 2.5 mm on MRI or CT evaluation.8
• Less than Grade 3 spondylolisthesis.8
• Not a surgical candidate or preference for non-surgical option.11
Multifactorial etiologies such as disc disease, facet hypertrophy, lateral recess/foraminal stenosis are not contraindicated as debulking the ligamentum flavum with mild has successfully treated LSS narrowing due to multiple etiologies in the clinical setting.12
Reimbursement
New CPT Category I codes for the mild procedure have been approved and will go into effect on January 1, 2026.13,14
62330 Decompression, percutaneous, with partial removal of the ligamentum flavum, including
laminotomy for access, epidurography, and imaging guidance (ie, CT or fluoroscopy), bilateral;
one interspace, lumbar
62331 additional interspace(s), lumbar (List separately in addition to code for primary procedure)
These codes will replace the current Category III code (0275T).15 This transition reflects the procedure’s established clinical utility, widespread adoption, and specialty society support. Category I status affirms that the procedure is no longer investigational and warrants standardized coverage and payment. We encourage payors to align coverage policies with this AMA determination to ensure timely and equitable access for eligible patients.
Conclusion and Recommendation
The mild procedure has proven to be a safe and durable option for the management of LSS with NC when identified using clinical examination and radiological findings of HLF ≥ 2.5 mm. Published evidence supporting mild include three level 1 RCTs, five prospective studies, two meta-analyses, four retrospective studies and three case series demonstrating the efficacy in decreasing symptoms of NC with significant improvements in physical functionality, and a strong safety profile similar to an ESI.6
NJSIPP strongly urges payors to adopt favorable coverage policies for the mild procedure in recognition of its clinical efficacy, cost-effectiveness, and alignment with the new Category I CPT® coding. Doing so will address a significant unmet need among patients suffering from debilitating spinal conditions and support broader access to evidence-based interventional pain care.
Respectfully,
Steve M. Aydin, DO, President NJSIPP
Sandeep Rathi, MD, Executive Director NJSIPP
Executive Board of the New Jersey Society of Interventional Pain Physicians
References
1 Mekhail N, Costandi S, Abraham B, Samuel SW. Functional and patient-reported outcomes in symptomatic lumbar spinal stenosis following percutaneous decompression. Pain Pract. 2012;12(6):417-425. doi:10.1111/j.1533-2500.2012.00565.x.
2 Deer TR, Kim CK, Bowman RG, Ranson MT, Yee BS. Study of percutaneous lumbar decompression and treatment algorithm for patients suffering from neurogenic claudication. Pain Physician. 2012 Nov-Dec;15(6):451-460.
3 Kalichman L, Cole R, Kim DH, et al. Spinal stenosis prevalence and association with symptoms: The Framingham Study. Spine J. 2009 Jul;9(7):545-550. doi: 10.1016/j.spinee.2009.03.005. [Epub 2009 Apr 23].
4 Jain S, Deer TR, Sayed D, et al. Minimally invasive lumbar decompression: a review of indications, techniques, efficacy, and safety. Pain Manag. 2020;10(5). https://doi.org/10.2217/pmt-2020-0037. Accessed June 1, 2020.
5 S. Costandi, T. Deer, T. Chafin, C. Kim. Three-year results of the MOTION randomized controlled trial for treatment of lumbar spinal stenosis using the percutaneous mild® Procedure. Interventional PainMedicine, Vol 4, Issue 1, 2025, 100561, ISSN 2772 5944,https://doi.org/10.1016/j.inpm.2025.100561
6 Benyamin RM, Staats PS. mild® is an effective treatment for lumbar spinal stenosis with neurogenic claudication: MiDAS ENCORE randomized controlled trial. Pain Physician. 2016 May;19(4):229-242.
7 Mekhail N, Costandi S, Nageeb G, Ekladios C, Saied O. The durability of minimally invasive lumbar decompression procedure in patients with symptomatic lumbar spinal stenosis: Long-term follow-up [published online ahead of print, 2021 May 4]. Pain Pract. 2021;10.1111/papr.13020.
8 Deer T, Grider J, Pope J et al. Best Practices for Minimally Invasive Lumbar Spinal Stenosis Treatment 2.0 (MIST): Consensus Guidance from the American Society of Pain and Neuroscience (ASPN). Journal of Pain Research 5 May 2022, p 1325-1354.
9 Udeh BL, Costandi S, Dalton JE, Ghosh R, Yousef H, Mekhail N. The 2-year cost-effectiveness of 3 options to treat lumbar spinal stenosis patients. Pain Pract. 2015 Feb;15(2):107-16. doi: 10.1111/papr.12160. Epub 2014 Jan 3. PMID: 24393198; PMCID: PMC4786075.
10 Wang JJ, Bowden K, Pang G, Cipta A. Decrease in health care resource utilization with mild. Pain Med. 2013 May;14(5):657-661. doi: 10.1111/pme.12117. [Epub 2013 Apr 11].
11 Staats PS, Dorsi MJ, Reece DE, Strand NH, Poree L, Hagedorn JM. Percutaneous image-guided lumbar decompression and outpatient laminectomy for the treatment of lumbar spinal stenosis: a 2-year Medicare claims benchmark study. Interv Pain Med. 2024 Apr 22;3(2):100412. doi: 10.1016/j.inpm.2024.100412. PMID: 39238588; PMCID: PMC11373052.
12 Deer T, Kim C, Wahezi SE, Qu H, Sayed D; MOTION Study Investigators. Objective Real-World Outcomes of Patients Suffering from Painful Neurogenic Claudication Treated with the mild® Procedure: Interim 6-Month Report of a Randomized Controlled Trial. J Pain Res. 2021 Jun 10;14:1687-1697. doi: 10.2147/JPR.S312573. PMID: 34140805; PMCID: PMC8203201.
13 AMA CPT Panel Meeting September 2024 Summary of Panel Actions
14 CPT Changes: An Insider's View 2026
15 Percutaneous laminotomy/laminectomy (interlaminar approach) for decompression of neural elements, (with or without ligamentous resection, discectomy, facetectomy and/or foraminotomy), any method, under indirect image guidance (eg, fluoroscopic, CT), single or multiple levels, unilateral or bilateral; lumbar
New Jersey State Legislative Update
Laurie Clark – NJSIPP Lobbyist
March 30, 2026
APN BILL SIGNED BY THE GOVERNOR
S-2996 / A-4052 – INDEPENDENT PRACTICE FOR APNs
The bill was substantially amended to only apply to certain aspects of primary care (excluding obstetrical care) and low acuity behavioral health. This legislation S-2996/A-4052 would allow an advanced practice nurse (“APN”) to provide health care services and prescribe medications to patients without physician collaboration or a written joint protocol with a collaborating physician ONLY in certain primary care settings and low acuity behavioral health services. Collaboration is required in all other medical specialties. In addition, the advanced practice nurse must have more than 5,000 hours of licensed, active, advanced nursing practice in a role with the applicable population focus. The amendment we advocated for increased the number of hours rom 2,400 to over 5,000. Although this is a step forward for the APNs, they are not happy and pledged to come back for all the specialties. We must keep up the fight.
The new law takes effect immediately, but regulatory changes are required to occur within 180 days of the bill’s signing.
We are greatly disappointed in this decision but we pledge to fight in a new way with enhanced data and regulatory input.
Sincere gratitude to all who assisted in our grassroots campaign
Please join us. More details to come.
New Jersey State Election Update
Laurie Clark – NJSIPP Legislative Counsel
Governor’s Race
On Tuesday, November 4, Congresswoman Mikie Sherrill was elected to be the 57th Governor of New Jersey. Top vote getters: The Governor – elect received 1, 829, 147 votes (56.5%) Jack Ciattarelli received 1, 392,136 votes (43.0%). The new Governor will be sworn in on January 20, 2026. We congratulate the new Governor-elect and look forward to working with the Governor and the new administration on critical healthcare issues.
Legislative Races
The New Jersey General Assembly will remain in Democratic control after Tuesday’s election. The members of the Assembly Democratic Caucus convened in Trenton yesterday to select the Leadership Team for the 222nd Legislative Session. Speaker Craig J. Coughlin was chosen to serve a historic fifth term as Speaker of the New Jersey General Assembly.
The Caucus met following Tuesday's election in which high turnout helped power Democratic victories across the state. Democrats in the Assembly successfully defended every seat and flipped an additional three seats for a 55 - 25 majority, with some races still not called. That is the largest majority held by Democrats in fifty years.
The gains in Tuesday's election show a continued voter shift to the Democratic Party. Since the 2023 election, Assembly Democrats picked up nine seats. The last time a party picked up four or more seats in consecutive elections was 1989-1991
The Caucus also voted to return Majority Leader Louis D. Greenwald (D-Camden, Burlington) to serve an eighth term in his leadership position. Speaker Pro Tempore Annette Quijano (D-Union) will begin her first full-term in this position, and Assemblywoman Linda Carter (D-Somerset, Union) will begin her first full term as Majority Conference Leader
Senate President Nick Scutari and Senate Majority Leader M. Teresa Ruiz were both unanimously reelected to their leadership positions for the 2026-27 legislative session by their Senate Democratic colleagues today.
Assemblyman John DiMaio was selected once again to be the Assembly Republican Leader.
Senator Anthony Bucco was re=elected as the Senate Republican Leader.
The members of the Senate Democratic Majority voted for the third time to have Senate President Scutari and Senate Majority Leader Ruiz continue in the top Senate posts they first assumed for the 2022-23 legislative session.
Reorganization of the 222nd Legislative Session will take place on January 13, 2026, when all members will be sworn in and an official vote on leadership will take place.
New Jersey Department of Banking and Insurance PIP Fee Schedule Revamp Looms
ICYMI – The NJSIPP Team was remarkably busy over the past few months. We responded to the proposal and met with key legislators. However, there is still a threat of this happening during the transition period between administrations.
We are continuing our work. Further updates will be forthcoming.
Senate Health Committee Considering Voluntary Non-Opioid Directives on November 13
The Legislature is back and the “Lame Duck Session” is officially underway. On Thursday, November 13, the committee has a full agenda including:
S4337/A5595 Allows individuals to establish voluntary nonopioid directives
Under the bill, an individual who is 18 years of age or older, an emancipated minor, or a patient’s authorized representative, as this term is defined in the bill, may execute a voluntary nonopioid directive stating that an opioid may not be administered or prescribed to the individual.
A voluntary nonopioid directive is to be made on a form that is to be developed by the Department of Health and published on the department’s Internet website. Nothing in the bill is to prevent an opioid from being prescribed to a patient if deemed medically necessary.
A health care professional, a health care facility, or an employee of a health care professional or health care facility is to be immune from disciplinary action by the Department of Health or a licensing agency or board for any act that was done to comply with the bill’s provisions. Further, a health care professional, a health care facility, or an employee of a health care professional or health care facility is to be immune from any civil or criminal liability for failure to administer, prescribe, or dispense an opioid, and for inadvertent administration of an opioid, to an individual who has a voluntary nonopioid directive, if the act or the failure was reasonable and done in good faith._____________________________________________________________________________________________________________________
NJSIPP Board Meeting with NJ Senate Leader Nick Scutari: Safeguarding Access to Care Amid PIP Fee Schedule Reform
October 15, 2025

Yesterday, the board of the New Jersey Society of Interventional Pain Physicians (NJSIPP) convened an important meeting with Senator Nick Scutari, Senate President and longtime champion of consumer protection and health care reform. The core focus: proposed changes to New Jersey’s Personal Injury Protection (PIP) medical fee schedule and ensuring that injured citizens retain access to high-quality care.
Key Highlights & Takeaways
1. Balancing cost control and patient access
Senator Scutari underscored the necessity of regulating medical reimbursement rates under PIP—but he also affirmed that any reform must avoid creating a “payment cliff” that would push providers to refuse PIP patients or refuse necessary services. The Senate leader emphasized that affordability for insurers cannot come at the expense of care providers’ viability or patient outcomes.
2. Protecting injured patients from balance billing
One of the strongest protections under discussion is a prohibition on balance billing for medical services tied to PIP claims. Under legislation such as S3963, unreimbursed medical expenses above PIP limits would fall under the same regulated fee schedule, and providers would be barred from demanding additional payment from patients.
3. Indexing and updating the schedule
We discussed a biennial inflation adjustment mechanism (e.g. cost-of-living, updates for new procedures) to keep the fee schedule responsive to real-world costs—so providers aren’t perpetually operating at a loss. Senator Scutari expressed openness to rule-making flexibility, especially for rural or specialty practices.
4. Stakeholder voices and transparency
NJSIPP representatives pressed for a transparent rule-making process, with sufficient public comment periods and input from providers, insurers, and advocacy groups. Senator Scutari pledged his support for an open, iterative approach rather than “last-minute” changes behind closed doors.
5. Ensuring continuity of care
Above all, the conversation returned again and again to the injured New Jerseyans who depend on PIP coverage to access rehabilitation, therapies, diagnostic imaging, and other critical services post-accident. It’s vital that reforms not inadvertently narrow provider networks or lead to care deserts for the most vulnerable.
✅ Why This Matters
- Patients: Without careful calibration, a lower reimbursement schedule may discourage participation from providers, limiting access for injured individuals.
- Providers & Clinics: Practices already operating on tight margins must absorb cost pressures; meaningful rate updates & inflation indexing are essential for sustainability.
- Insurers & Regulators: A transparent, predictable framework supports better rate-setting and risk management.
- Policymakers & Advocates: This board meeting is a model for constructive policymaking—bringing real-world professionals, legislators, and public-interest voices together before rules are finalized.
Call to Action:
- If you are a provider, insurer, patient advocate, or legislative staffer concerned about PIP reform in New Jersey, I encourage you to:
- Monitor upcoming rule-making hearings
- Submit comments or testimony during public notice periods
- Engage your State Senator and Assembly member
- Collaborate through NJSIPP or allied professional associations
- Let’s make sure PIP reform strengthens, rather than weakens, access to care in New Jersey
________________________________________________________________________________________________________
NJSIPP LEGISLATIVE UPDATE
July 1, 2025
Laurie Clark – Legislative Counsel
GOVERNOR MURPHY SIGNS FISCAL YEAR 2026 BUDGET INTO LAW
Ambulatory Surgery Center Tax Increase– Effective July 1
Despite our best efforts to stop it, the elimination of both the current $300,000 floor and the $350,000 CAP on the amount of the tax was passed in a standalone bill and signed into law on June 30. The new law is targeted to save $70 million over this fiscal year. Once the Legislature returns to session, coalition efforts will begin to attempt to change the assessment to net rather than gross receipts.
Known as the “Healthcare Finance Enhancement Act” (Assembly Bill 5809), the new law brings significant changes to the assessment of ambulatory care facilities, including ambulatory surgery centers, surgical practices, and licensed imaging centers (ACFs). Prior to the passage of the new law, ACFs were required to pay a 2.95% assessment on gross receipts over $300,000, with a cap on the assessment of $350,000. Under the new law, beginning July 1, 2025, the assessment is set at 2.5% of gross receipts, and both the floor of $300,000 and the cap of $350,000 have been removed. Further, while certain facilities, such as one (1) room surgical practices, were previously exempt from the assessment, these facilities will now be subject to the same assessment.
These changes will impact almost every ACF in New Jersey. While many facilities will have a reduced financial burden due to the decrease in the assessment percentage, many other facilities, such as surgical practices, will no longer be exempt from an assessment. Additionally, facilities that generate larger gross receipts will be assessed higher amounts, as the $350,000 cap has been lifted, and facilities that generate gross receipts under $300,000 will no longer be exempt.
The $58.78 billion Fiscal Year 2026 (FY2026) budget, which was passed by the Legislature and signed by the Governor, redirects over 75 percent of the total budget back into local communities in the form of grants-in-aid for property tax relief, social services, higher education, as well as State aid to schools, municipalities, and counties.
FY26 Budget Highlights
- Final FY26 Budget by the Numbers
-
- State appropriations: $58.78 billion
- State revenue: $57.31 billion
- Surplus: $6.7 billion
- Spending increase from FY25 to FY26: 4%
- Spending increase from budget proposed by Legislature in February to final budget in June: $767 million or 1.3%
- 5th straight year of 100% pension payment
The Legislature is expected to return to session in November.
________________________________________________________________________________________________________
UPDATE: GOVERNOR MURPHY’S NJ STATE FISCAL YEAR 2026 BUDGET
I attended in person, the Governor’s Budget Message and the Commissioner of Human Services, Medicaid Briefing.
For now, the proposed $58.1 billion budget is presented “status quo” without large programmatic cuts. However, the Governor did not rule out a “break the glass” emergency.
As you can see from the news coming out of Washington, DC, the House and US Senate passed different budget bills. There are also varying statements concerning the Medicaid/Medicare funding.
The Federal Medicaid funding is in danger, and everyone was encouraged to stay in contact with their representatives in Congress. New Jersey currently receives 50% federal match on Medicaid spending from the federal government. March 14 will be a pivotal day in Washington, DC as the final Congressional Resolution (CR) to fund the government as well as the debt ceiling must be re-authorized by Congress.
This state budget provides $6.3 billion for Medicaidmich for which we receive the same amount in federal matching funds. Currently, 1.8 million individuals utilize these programs.
Ambulatory Surgery Center Tax Changes
There are changes to the Ambulatory Surgery Center Tax that will negatively impact the owners of larger centers. Governor Murphy is proposing aligning the ASC tax rates with federal Medicaid Provider taxes allowing a reduction in the nominal rate from 2.95% to 2.5% but eliminating the cap. This is estimated to raise more than $60 million in new state revenues.
Primary Care Funding for Medicaid/Medicare Parity
An increase in current Medicaid Reimbursement for Primary Care. Including a new $49 million appropriation.
Attracting more OB-GYN’s
A new program to attract more OB-GYNs to New Jersey and retain the existing workforce is accomplished by increasing reimbursement rates.
Hospital Funding
Hospitals will receive $3.6 billion in direct state funding under this proposed budget. This represents $2.9 billion in increased funding since Governor Murphy took office.
Graduate Medical Education Funding
The proposed budget includes $336 million in funding including the supplemental and trauma components
This a partial list of some of the major healthcare components of the proposed budget. More will be provided in the next update. The Legislative review of the budget begins now. We will be fighting all elements of the budget that negatively impact on patient care….
Renew your membership and donate to PAC!
New Jersey State Legislative Update
Laurie Clark – Legislative Counsel to NJSIPP
February 13, 2025
We started the New Year with positive news on the legislative and regulatory fronts!
Telemedicine Parity Extension Signed into Law
Bill Was Signed on December 31
On December 19, both Houses of the Legislature voted to pass S2988/A3853 sponsored by Senator Vin Gopal and Assemblyman Herb Conaway.
This bill amends section 11 of P.L.2021, c.310 to extend the end date from December 31, 2024 to July 1, 2026 during which time a health benefits plan in this State is to provide coverage and payment for health care services delivered to a covered person through telemedicine or tele-health at a provider reimbursement rate that equals the provider reimbursement rate that is applicable, when the services are delivered through in-person contact and consultation in New Jersey, provided the services are otherwise covered by the health benefits plan when delivered through in-person contact and consultation in New Jersey.
The bills were signed into law on December 31. The law became effective immediately and extends the pay parity regulations for New Jersey Insurance Coverage until July 1, 2026.
Please note that Medicare Regulations on Telehealth Pay Parity have been extended until March of 2025. We await further Congressional action in the new session which begins tomorrow.
New Jersey Department of Banking and Insurance Issues Bulletin on Prior Authorization Law
On December 31, 2024, the New Jersey Department of Banking and Insurance issued several key bulletins effectuating the provisions of laws signed in the previous legislative session (2023). These Bulletins have the full force of law and are mandatory for compliance by insurance carriers operating in New Jersey beginning on January 1, 2025.
Please review the bulletin concerning prior authorization at the link below.
We are especially happy about the provisions that require a physician of the same specialty be directly involved with the internal appeal. Our society worked very hard on those provisions.
Personal Injury Protection Auto Insurance Reforms Underway
On Monday, the Senate Commerce Committee chaired by Senator Joseph Lagana reported favorably and with committee amendments Senate Bill No. 1473 (Lagana/Bramnick).
As amended, this bill revises the personal injury protection coverage for basic automobile insurance policies from $15,000 to $20,000 and requires a minimum personal injury protection coverage of $50,000 for standard automobile liability insurance policies. Under the bill, every basic automobile insurance policy will be required to provide personal injury protection coverage in an amount not to exceed $20,000 per person per accident, and every standard automobile liability insurance policy will be required to provide personal injury protection coverage that shall be no less than $50,000 per person per accident. Current law requires basic automobile insurance policies to provide personal injury protection coverage in an amount not to exceed $15,000 per person per accident, with no minimums required for standard automobile liability insurance policies. Additionally, the bill requires insurers to inform a named insured that policy limits for the insured’s standard automobile liability insurance policy or basic automobile insurance policy have increased during the renewal period of the policy but exempts insurers from needing a signed coverage selection form before increasing these limits.
This bill was pre-filed for introduction in the 2024-2025 session pending technical review. As reported, the bill includes the changes required by technical review, which has been performed.
COMMITTEE AMENDMENTS:
The committee has amended the bill to:
(1) revise personal injury protection coverages for basic automobile insurance policies from $50,000 to $20,000;
(2) require insurers to inform a named insured that policy limits have increased when the named insured has renewed a policy of insurance and exempt insurers from receiving a signed coverage selection form before increasing a policy’s limits;
(3) update current law reviewing personal injury protection options for standard automobile liability insurance policies;
(4) revise the effective date to require that the revised personal injury protection coverages for basic automobile insurance policies be implemented on the first day of the 18th month after the date of enactment of this bill; and
(5) make certain technical changes.
The bill is now on second reading and ready for a Senate floor vote.
In addition, work on a new PIP Fee Schedule is in progress. The goal is to have it in place by the end of 2025.
NJSIPP Executive Director Andy Kaufman, MD was a featured panelist at BioNJ’s Patient Advocacy Forum on December 13. The topic of the panel was “The Impact of Pharmacy Benefit Management Practices on Patients”
Dr. Kaufman ‘s participation was highly acclaimed and of great assistance to the patient advocates in attendance.
|
|
|
Dr. Woska being honored for his service to our society as the Executive Director for the past 5 years. We congratulate him for all the time and effort that he put forth and the excellent job that he did.
-
Proposed Medicare National Coverage Determination for Epidural Steroid Injection
Effective 12/05/2021
The link to the policy is available here:
https://www.cms.gov/medicare-coverage-database/view/lcd.aspx?lcdid=38994&ver=7&bc=0
Covered Indications
1. Epidural steroid injection (ESI) will be considered medically reasonable and necessary when the following three (3) requirements are met:
-
History, physical examination, and concordant radiological image-based diagnostic testing and supporting one of the following:
Cervical, Thoracic, or Lumbar radiculopathy, radicular pain and/or neurogenic claudication due to central disc herniation, osteophyte or osteophyte complexes, severe degenerative disc disease, producing foraminal or central spinal stenosis.
OR
Post-laminectomy syndrome
Acute herpes zoster associated pain
AND
Symptoms severe enough to impact quality of life or function. An objective pain scale or functional assessment must be performed prior and repeated following intervention to assess response.
AND
Pain duration of at least 4 weeks, and in the inability to tolerate noninvasive conservative care or failure to respond to 4 weeks of noninvasive conservative care.
*Acute herpes zoster pain refractory to conservative care, 4 week wait is not required
2. ESI must be performed under CT or fluoroscopy image guidance with contrast,unless the patient has a documented contrast allergy or pregnancy where ultrasound guidance without contrast may be considered.
3. Transforaminal EPIDURAL steroid injections (TFESIs) involving a maximum of two (2) levels in one spinal region are considered medically reasonable and necessary. It is important to recognize that most conditions would not ordinarily require ESI at two (2) levels in one spinal region.
4. Caudal EPIDURAL steroid injections (CESIs) and interlaminar EPIDURAL steroid injections (ILESIs) involving a maximum of one level are considered medically reasonable and necessary.
5. It is medically reasonable and necessary to perform TFESIs bilaterally only when clinically indicated.
6. Repeat ESI when the first injection:
At least 50% sustained improvement and/or improvement in function.
If patient fails to respond, repeat ESI after 14 days can be performed, using a different approach, level and/or medication if appropriate, with rationale and medical necessity for second ESI documented in EMR.
7. An initial injection of contrast is required to confirm epidural placement unless the patient has a contraindication to contrast. The subsequent epidural steroid injections should include corticosteroids and may be combined with anesthetics or saline.
8. The ESIs should be performed in conjunction with conservative treatments.
9. Patients should be part of an active rehabilitation program, home exercise program or functional restoration program.
*Note: The scales used to measure of pain and/or disability must be documented in the medical record. Acceptable scales include but are not limited to: verbal rating scales, Numerical Rating Scale (NRS) and Visual Analog Scale (VAS) for pain assessment, and Pain Disability Assessment Scale (PDAS), Oswestry Disability Index (ODI), Oswestry Low Back Pain Disability Questionnaire (OSW), Quebec Back Pain Disability Scale (QUE), Roland Morris Pain Scale, Back Pain Functional Scale (BPFS), and the PROMIS profile domains to assess function.
Limitations
1. Injections performed without image guidance or by ultrasound are not considered reasonable and necessary except in cases of documented contraindication to contrast media (e.g., allergy, pregnancy)
2. ESIs performed with biologicals or other substances not FDA designated for this use is considered investigational and not medically reasonable and necessary
3. It is not considered medically reasonable and necessary to perform multiple blocks (ESI, sympathetic blocks, facet blocks, trigger point injections etc.) during the same session as ESIs, with the exception of a facet synovial cyst and ESI performed in the same session
4. Use of Moderate or Deep Sedation, General Anesthesia, and Monitored Anesthesia Care (MAC) is usually unnecessary or rarely indicated for these procedures and therefore not considered medically reasonable and necessary.Even in patients with a needle phobia and anxiety, typically oral anxiolytics suffice. In exceptional and unique cases, documentation must clearly establish the need for such sedation in the specific patient.
5. ESIs to treat non-specific low back pain (LBP), axial spine pain, complex regional pain syndrome, widespread diffuse pain, pain from neuropathy from other causes, cervicogenic headaches are considered investigational and therefore are not considered medically reasonable and necessary.
6. ESIs are limited to a maximum of four (4) sessions per spinal region in a rolling twelve (12) month period.
7. It is not considered medically reasonable and necessary for more than one spinal region to be injected in the same session.
8. It is not considered medically reasonable and necessary to perform TFESIs at more than two (2) nerve root levels during the same session.
9. It is not considered medically reasonable and necessary to perform CESIs or ILESIs at more than one (1) level during the same session.
10. It is not medically reasonable and necessary to perform CESIs or ILESIs bilaterally.
11. It is not medically reasonable and necessary to prescribe a predetermined series of ESIs.
12. Steroid dosing should be the lowest effective amount, it is recommended not to exceed 80mg of triamcinolone, 12 mg of betamethasone, 15 mg of dexamethasone per session.
13. It generally would not be considered medically reasonable and necessary for treatment with ESI to extend beyond 12 months.Frequent continuation of epidural steroid injections over 12 months may trigger a focused medical review. Use beyond twelve months requires the following:
a. Pain is severe enough to cause a significant degree of functional disability or vocational disability.
b. ESI provides at least 50% sustained improvement of pain and/or 50% objective improvement in function (using same scale as baseline).
c. Rationale for the continuation of ESIs including but not limited to patient is high-risk surgical candidates, the patient does not desire surgery, recurrence of pain in the same location relieved with ESIs for at least three months
d. The primary care provider must be notified regarding continuation of procedures and prolonged repeat steroid use.
14. ESIs should not be performed when contraindicated including but not limited to: suspected or active localized spinal infection, significant systemic infection, compressive lesions of the spinal cord, conus medullaris or cauda equina, suspicion or major risk factors for cancer.
Update on New Medicare Facet Rules
Dr. Steve Aydin created this informative presentation that was shared during the NJSIPP Annual Fall Meeting. We thank him for sharing his slides below.
URGENT - Medicare proposed reduction in sacral and knee RF reimbursement
This topic was discussed the NJSIPP meeting on Wednesday night. It is Urgent you respond - there are only 10 days left!!!
Medicare has proposed to significantly decrease the payment rates for both the physician and the facility setting for the new knee and SIJ radiofrequency ablation CPT codes that are effective January 1, 2020. These new codes will impact all RFA technologies, both cooled and conventional.
We encourage you to:
1. review the proposed rules and provide comments to Medicare. The deadline for comments is September 27, 2019. Links to the proposed rules and information on how to submit comments are noted at the end of this e-mail. It is very important that Medicare receives comments from as many stakeholders as possible before the Final Rules are released later this year.
2. connect with your societies who participated in the new CPT code development process to express your concerns and encourage them to comment on the proposed rules as well.
Here are some important key facts from the proposed rules:
KNEE RFA IMPACTS
- Physician Rule – The proposed physician RVU valuation for the new CPT code for RFA in the knee has significantly decreased from the current peripheral nerve ablation RVUs. Avanos believes the reduction in rate is not appropriate and will likely result in a decrease in usage of this non-opioid alternative to treating pain. Avanos plans to raise this issue in its comment letter.
- Physician & HOPD Rules – The new Knee RFA CPT code was developed to report 3 nerve ablations during a single procedure versus the current peripheral nerve ablation code where reporting is on a per ablated nerve basis.
- HOPD Rule – The hospital outpatient facility payment for the knee RFA procedure has significantly decreased. The new knee RFA CPT code was assigned to the Nerve “Injection” APC (Ambulatory Payment Classification) unlike the new SIJ procedure CPT code which was assigned to the Nerve “Procedure” APC. Avanos believes that the knee RFA procedure should be properly placed in the Nerve Procedure APC and intends to address this in its comment letter.
The following table provides a snapshot of the proposed changes in Medicare National average payment rates:
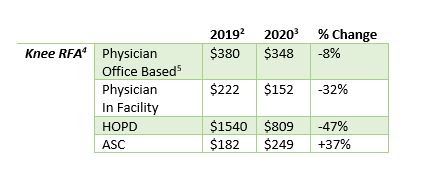
SIJ RFA IMPACTS
- Physician Rule – The proposed physician RVU valuation for the new code for SIJ RFA procedures has significantly decreased from the current peripheral nerve ablation RVUs. Avanos believes the reduction in rates is not appropriate and will likely result in a decrease in usage of this non-opioid alternative to treating pain. Avanos plans to raise this issue in its comment letter.
- Physician & HOPD Rules – The new SIJ RFA CPT code was developed to report 3 nerve ablations during a single procedure versus the current peripheral nerve ablation code where reporting is on a per ablated nerve basis.
- The following table provides a snapshot of the proposed changes in Medicare National average payment rates.
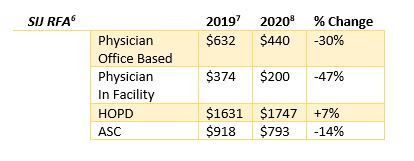
Submit your online comments related to CMS Proposed HOPD:
Submit electronic comments on this HOPD regulation to:
Click on - Regulations.gov – CMS-1717-P
or
https://www.regulations.gov/searchResults?rpp=25&po=0&s=CMS-1717-P&fp=true&ns=true
Follow the instructions under the “Comment Now” tab. Reference Rule CMS-1717-P.
Submit your online comments related to CMS Proposed Physician:
Submit electronic comments on this Physician regulation to:
Click on - Regulation.gov – CMS-1715-P
or
https://www.regulations.gov/searchResults?rpp=25&po=0&s=CMS-1715-P&fp=true&ns=true
Follow the instructions under the “Comment Now” tab. Reference Rule CMS-1715-P.



